Sponsored content
The signing of the 2018 Farm Bill – which legalized the growth and manufacturing of industrial hemp – generated an explosion of products available to the consumer. Since the passing of that bill and up to today, there are no consistent regulations for how industrial hemp products, currently focused on cannabidiol (CBD) content, are manufactured and marketed, according to Golden, Colorado-based Panacea Life Sciences. 
The lack of rules and regulations has created quality issues as demonstrated in the June 2019 FDA Meeting where 70 percent of products failed to meet quality control standards, thus leading to consumer and retail confusion. One of the current misconceptions is regarding claims of products being Tetrahydrocannabinol (THC)-free. When labeled as such, CBD consumers are led to believe they would not experience psychogenic effects while attaining other health benefits from CBD, and furthermore, the claim would ensure that these products will not cause individuals to test positive for THC use. Gaining a greater understanding of how these products are made and why there has been a recent change in reported THC levels will allow retailers and consumers to make informed decisions on which products they should choose for maximum health benefits, and if needed, to minimize THC consumption.
Industrial hemp plant processing
The industrial hemp plant, a sub-breed of Cannabis sativa L., has been selectively bred to have high cannabidiol (CBD) content and low THC levels. To be classified as industrial hemp, the THC levels must be below 0.3 percent. These plants contain a rich mixture of more than 500 distinct compounds reported to provide numerous health benefits. Of these compounds, there are more than 113 identified cannabinoids, two of which are becoming more well-known to the general consumers: CBD and THC.
Cannabidiol is a non-psychogenic cannabinoid possessing many reported biological assets including anti-inflammatory and antioxidant properties. THC is a psychogenic cannabinoid that produces the euphoric high associated with cannabis use. Throughout the process of growing and manufacturing hemp products, the THC level is constantly tested to ensure the THC content is compliant with federal law.
While each state where hemp is grown will have slightly different rules and procedures for farming hemp, in Colorado, growers work closely with the Department of Agriculture (CDA) to register each plot of land where hemp will be planted. The plants are randomly tested by the CDA to ensure that the THC content is less than 0.3 percent before permission is granted to harvest the plant. Non-compliant crops are destroyed, and growers face potential fines and limitations on the ability to plant hemp in the following season.
Once the hemp is harvested, the crop is transferred to an extraction facility where, through a variety of methods, the oil is separated from the plant material. The most commonly used methods are ethanol or carbon dioxide extraction. In each approach, a solvent (ethanol or liquid CO2) is mixed with hemp plant material to solubilize the oil. The plant material is then separated from the oil by filtration, and the solvent is removed to create CBD-rich hemp oil. The oil can be further refined through distillation, at which point the CBD can be enriched to create CBD-isolate through selective precipitation and filtering. This creates a solid powder of >97% purity that looks much like powdered sugar. It can also be refined by using chromatography which removes the THC from the oil to create “THC-FREE,” or broad spectrum, distillate.
Different forms and definitions of CBD ingredients
Manufacturers will utilize three main CBD ingredients to create finished goods — each of which will appeal to different consumers:
Full Spectrum Oil: This is hemp oil that contains all of the components of the plant including minor cannabinoids, terpenes and flavonoids. These products will contain THC that should not exceed 0.3 percent. The terpenes and flavonoids give hemp oil its marijuana smell and taste and offer additional health benefits.
Consumers choose full spectrum oil because of the entourage effect, where the multiple components in the help oil work synergistically to reportedly provide greater health benefits than CBD alone. Consumers who do not like the earthy taste of hemp oil or are concerned about testing positive for THC use should choose a product that has the THC removed.
Broad Spectrum Oil: This is oil that has been processed to remove as much THC as possible while keeping as many of the other cannabinoids and terpenes in the oil. This allows consumers to benefit from the entourage effect while minimizing any THC exposure.
CBD isolate: This is pure CBD (typically over 97 percent pure) that has almost all of the other components of CBD removed and is a tasteless and odorless white powder. Consumers may choose isolate products to receive the anti-inflammatory benefits of CBD in a pleasant tasting product that minimizes THC exposure. This is a very versatile ingredient that manufacturers can incorporate into foods, edibles, tinctures and tablets without substantially altering a taste profile.
THC-FREE vs. THC-non-detect
The hemp/CBD industry has only legally been in existence for two years. Initially, testing laboratories employed a methodology that was geared to ensure products and plants met the 0.3 percent THC federal guidelines. This meant that the methods would have a limit of quantification (LOQ), the concentration that could be meaningfully determined, of approximately 0.2 percent THC, which is 2000 parts per million, or two parts per thousand. Two parts per thousand is roughly equivalent to adding two drops of solution into a quart of water. Any product that contains less than this level is reported as having a THC concentration of lower than zero. Manufacturers were reporting based on the information provided by the testing laboratory, so products that returned a value of zero THC could be considered THC-Free.
As the industry has rapidly evolved, testing laboratories have employed more sensitive methods such that the LOQ levels are now 0.01 percent or 100 ppm. This is equivalent to adding a drop of solution to two bathtubs full of water. Recognizing that there are trace amounts of THC that will be present in previously reported THC-FREE products, states like Colorado are in process of defining such products as THC-ND (non-detect) where THC levels must be lower than 100 ppm, or 0.01 percent.
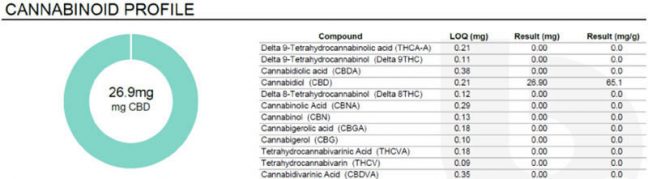
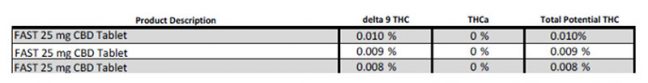
What does this mean for products? Take Panacea’s 25mg F.A.S.T. tablets for example. Before March 2020, this product would have a testing profile that showed below reporting 0 percent THC. The level of detection or quantification for many laboratories at this point was 0.2 percent or 2000 ppm. When individual tablets were evaluated using more sensitive THC quantification methods with an LOQ of 0.005 percent, it can be seen that the tablets do have trace amounts of THC ranging from 80 ppm to 130 ppm, or 0.03mg to 0.0mg THC per tablet. Therefore, under Colorado’s proposed definitions, this product would be labeled as a THC non-detect product.
2019 Cannabinoid Potency Test for Panacea Life Sciences FAST Tablets
Why the concern about THC concentrations?
Consumers are typically worried about the levels of THC in their products for two main reasons: they do not want the inebriation caused by THC, and/or they are concerned about testing positive for THC use.
While each individual responds to THC differently, a dose of 5mg of THC has been reported to get people “high.” The quantity of THC needed to achieve inebriation will vary based on body weight, dosage, method (smoking, oral, tincture), prior use of THC and other non-predictable sensitivities to THC. A 400mg tablet made from full spectrum hemp oil having a legally compliant 0.3 percent THC level would contain approximately 1.2mg THC per tablet. Using the 5mg threshold for inebriation would mean that those consuming 4-5 tablets could potentially get “high.” In contrast, using the example product of F.A.S.T. from above, a person would need to consume 125 F.A.S.T. tablets that contain the non-detect level of 0.04mg THC per tablet, all at one time, which is very unlikely.
For those employed in positions that require routine drug testing, such as first responders, federal employees including armed forces members, or professional athletes, the THC level in products is very important. Using a standard employer urinalysis test, a person would test positive for THC at urine levels greater than 50 ng/mL. Using a worst-case calculation to err on the side of caution, we will assume that 50 percent of the THC present in a product like F.A.S.T. is absorbed (termed bioavailability, and for most products is typically 10 percent of the product dose; this is being studied by Panacea Life Sciences in a pending human clinical trial). The average human urine output is 800mL (can be as high as 2L), and 20 percent of the THC consumed is eliminated in the urine, so the total amount of THC an individual would need to consume to test positive for THC use is 0.5mg per day. Again, using the F.A.S.T. tablet example, this would mean that a subject would need to consume more than 12 F.A.S.T. tablets to even potentially test positive in a drug test. This is six-fold higher than the daily recommended serving size of F.A.S.T.
In a one-person controlled trial where the patient consumed four F.A.S.T. tablets daily for 30 continuous days, a urinalysis tested negative for THC levels. These results are being expanded with more patients and dosages to provide greater consumer confidence regarding drug tests and THC non-detect products.

Regulations will increase consumer confidence and knowledge
As the Food and Drug Administration and individual states like Colorado institute consistent regulations regarding production and labeling, the definitions will become more uniform to give both consumers and retailers a better understanding of CBD oil products. It is equally important to standardize the THC level needed to qualify a product as either THC-Free or THC non-detect.
The overall goal is to raise consumer confidence that these products will not cause inebriation or show as positive for THC on a drug test. Doing so allows consumers to reap the benefits of CBD products without the potentially negative consequence of having trace amounts of THC in their bodies.